


The Long Road to a New Medicine
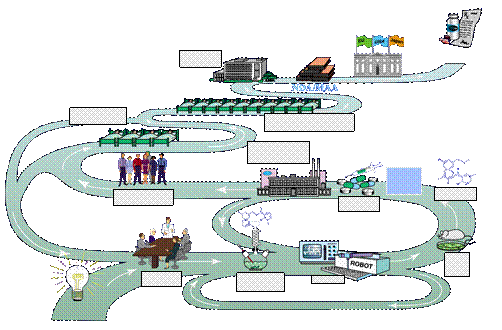
Discovery
Exploratory Development
Full
Development
Registration
Large
Amounts of
Candidate
Medicine
Synthesized
Project
Team
and
Plans
Synthesis
of
Compounds
Early
Safety
Studies
Candidate
Formulations
Developed
Extensive
Safety
Studies
Screening
Studies
in Healthy
Volunteers
Phase I
Candidate
Medicine Tested in
3-10,000
Patients (Phase III)
Studies
in 100-300
Patients
(Phase II)
Clinical
Data
Analysis